Evaluation of Bactericidal Activity of Electrochemical GO Modified with TiO2 Nanoparticles
Evaluación de la actividad bactericida de GO electroquímico modificado con nanopartículas de TiO2
Received: June 22, 2023
Accepted: December 23, 2023
Available: December 29, 2023
G. Durango Giraldo, J.C. Zapata-Hernández, C. Betancur Henao, J.F. Santa Marín, R. Buitrago Sierra “Evaluation of Bactericidal Activity of Electrochemical GO Modified with TiO2 Nanoparticles,” TecnoLógicas, vol. 26, nro. 58, e2765, 2023 . https://doi.org/10.22430/22565337.2765
Highlights
Highlights
Abstract
Recently, antibacterial materials have sparked a renewed interest in the fields of biomedical engineering and life sciences. The main purpose of this study was to evaluate the physicochemical properties of TiO2 nanoparticles with anatase phase and an average size of 24.1 ± 4.6 nm, graphene oxide (GO) obtained from the electrochemical method, and TiO2/GO hybrid nanomaterial. Thermogravimetric analysis (TGA) revealed the presence of oxygen functionalities in the GO structure and 23.2 % of TiO2 in the hybrid nanomaterial, as well as a strong interaction between the materials that can be observed in the micrograph of scanning electron microscopy (SEM). Antibacterial tests were performed using the macrodilution method. The results showed that, while GO did not decrease bacterial growth, TiO2 presented high bactericidal activity. In turn, the hybrid TiO2/GO nanomaterial did not show such activity. This result can be explained by the decrease in contact between TiO2 and bacterial cells due to the blocking of the active sites on the TiO2 surface by graphene oxide sheets. These results contribute to the ongoing discussion about the bactericidal properties of graphene oxide.
Keywords: Bactericidal activity, hybrid nanomaterial, graphene oxide, titanium dioxide.
Resumen
Recientemente, los materiales antibacterianos han despertado un renovado interés en el campo de la ingeniería biomédica y las ciencias de la vida. El objetivo de este estudio consistió en evaluar las propiedades fisicoquímicas de nanopartículas de TiO2 fase anatasa y un tamaño medio de 24.1 ± 4.6 nm, óxido de grafeno (GO) obtenido a partir del método electroquímico y un nanomaterial híbrido TiO2/GO. El análisis termogravimétrico (TGA) reveló la presencia de funcionalidades de oxígeno en la estructura del GO, y se encontró un 23.2 % de TiO2el nanomaterial híbrido y una fuerte interacción entre los materiales que puede observarse en las micrografías de microscopia electrónica de barrido (SEM). Las pruebas antibacterianas fueron realizadas usando el método de macrodilución. Los resultados evidenciaron que, mientras que el GO no disminuyó el crecimiento bacteriano, el TiO2 presentó una alta actividad bactericida. A su vez, el nanomaterial híbrido TiO./GO no mostró dicha actividad. Este resultado puede explicarse por la disminución del contacto entre el TiO2 y las células bacterianas debido al bloqueo de los sitios activos en la superficie del TiO2 por las láminas de óxido de grafeno. Estos resultados contribuyen a la discusión en curso sobre las propiedades bactericidas del óxido de grafeno.
Palabras clave: Actividad bactericida, nanomaterial híbrido, óxido de grafeno, dióxido de titanio.
1. INTRODUCTION
Health-care-associated infections, which are acquired during hospital stays, could represent a serious risk to human health [
Accordingly, the general interest in developing antibacterial materials has grown in recent years. Hybrid nanomaterials have potential in this regard because they facilitate the use of nanostructured materials with specific properties and excellent mechanical properties, as well as improved biocompatibility and antibacterial activity. This makes them good prospects in tissue engineering [
On the one hand, graphene oxide (GO) has attracted special attention because it is a carbon atom monolayer that forms a dense honeycomb structure containing carboxylic groups on the edges, as well as hydroxyl and epoxide groups on its two accessible sides. GO has excellent physicochemical properties, large specific surface, mechanical resistance, electrical conductivity, and stability in water [
Nowadays, Hummers’ method is the most widely implemented strategy to synthesize GO used in bacterial applications [
On the other hand, titanium dioxide (TiO2) is considered a suitable material for medical applications due to its low toxicity, excellent thermal properties, and chemical stability. Additionally, TiO2 exhibits antibacterial activity against E. Coli due to oxidative damage to the cell membrane [
The main objective of this study was to evaluate the antibacterial activity of synthesized graphene oxide (GO), titanium dioxide (TiO2) and a TiO2/GO hybrid. The combination of two types of nanoparticles with different morphologies and dimensions is a new way to produce functional hybrid materials with a synergistic improvement in material performance.
Recently, several hybrids composed of GO and conventional semiconductors demonstrated significantly enhanced photocatalytic performance during pollutant degradation. Specifically, TiO2/GO is a hybrid that could be considered as a promising material photocatalysis and hydrogen evolution [
Escherichia coli was the model bacteria used here to evaluate the bactericidal effect of the three materials. This study was conducted to contribute to the ongoing discussion about the antibacterial properties of GO. In addition, to the best of the authors’ knowledge, the antibacterial properties of GO synthetized by the electrochemical method had not been reported before. The electrochemical method can be used as an alternative to produce GO because it is sustainable and more environmentally friendly.
2. METHODOLOGICAL ASPECTS
2.1 Materials
All the materials employed here were reagent-grade and used without any further purification. Titanium isopropoxide (Ti[OCH(CH3)2]4, 97 %, Sigma-Aldrich), ethanol (99.5 % J.T Baker), high-purity graphite rods (3x305 mm, 99.9 %, SPI supplies), sulfuric acid (H2SO4, 96 %, Fisher Scientific) and sodium sulfate (Na2SO4, 99 %, Chemi) were used for the synthesis of TiO2 and graphene oxide. All the solutions were prepared using deionized water.
2.2 Methods
2.2.1 TiO2 synthesis
TiO2 nanoparticles were synthesized by the hydrothermal method [
2.2.2 GO synthesis:
GO was synthesized by the electrochemical exfoliation method. An electrolyte solution of Na2SO4 at 0.1 M was prepared in deionized water and the pH was adjusted (~6.5 to 7) with H2SO4. Then, two graphite rods were immersed in the electrolyte solution using a working distance of 20 mm, and they were connected to a DC voltage source (10 V). Finally, the product was washed several times with water and ethanol and dried in an oven at 80 °C.
2.2.3 TiO./GO composite synthesis
The TiO2/GO composite was prepared mixing the materials previously synthesized in a 1:3 wt. % ratio using a Q500 Sonicator (20 kHz, Qsonica LLC, USA). The ultra-sonication probe tip was immersed directly in the suspension, and the supplied energy was adjusted at 30 % of the maximum capacity in order to avoid heating problems. Ultrasound pulses were applied 1 s ON and 2 s OFF during 15 minutes. Finally, the sample was filtered and dried in an oven at 80 °C.
2.2.4 Antimicrobial test
The antimicrobial activity of TiO2,GO and the TiO2/GO hybrid nanomaterial was evaluated using the macrodilution method, as described in our previous paper [
 (1)
(1)
Where sample OD is the absorbance of each sample tested with the material dilutions, and control OD is the optical absorbance of the untreated sample.
The Minimum Inhibitory Concentration (MIC) required to inhibit the growth of 50 % of organisms (MIC50) was calculated using and the selection criterion was R2 ≥ 0.99.
2.3 Physical-chemical characterization
The phase identification and lattice structure characterization of the synthesized TiO2 were performed using an X-ray diffractometer (PANalytical Empyrean Series II), operated with Cu Kα radiation (λ = 1,540 Å). The thermal stability was characterized by thermogravimetric analysis (TGA) using a discovery 550 Thermogravimetric Analyzer with a heating program, initially, under a nitrogen atmosphere up to 800 °C. Subsequently, the atmosphere was changed to air. The functional groups present in the samples were analyzed by infrared spectroscopy in a IRTracer-100 spectrophotometer with wavelengths between 500 and 4000 cm-1. The numbers of defects and layers in the GO were estimated by a LabRAM HR Raman spectrometer (Horiba-Jobin Yvon). [
3. RESULTS AND DISCUSSION
Figure 1a and 1b show the X-ray diffraction pattern of TiO2 nanoparticles and the Raman spectra of graphene oxide, respectively. The XRD pattern (Figure. 1a) shows anatase-phase TiO2 with characteristic diffraction peaks of 20 values at about 25.3°, 37.8°, 48°, 53.9° and 55°, they are attributed to the (101), (004), (200), (105) and (211) planes, respectively. These results are in agreement with those reported by. [
Figure 1b shows the Raman spectra of graphene oxide, with three characteristic bands: D-band (~1339 cm-1), G- band (~1583 cm-1) and 2D-band (~2686 cm-1). The G-band represents the sp2 hybridization of carbon atoms [
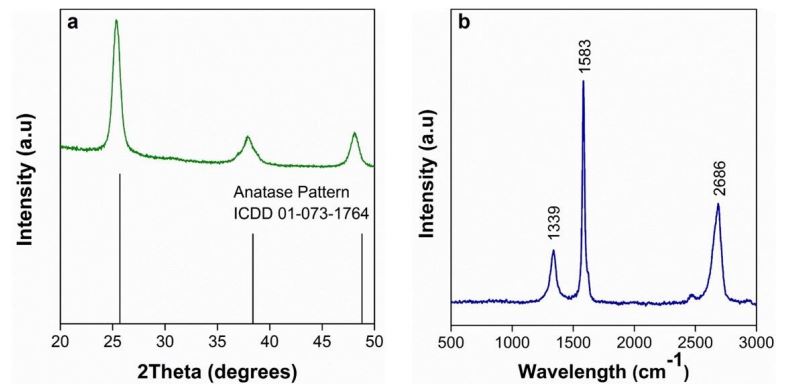
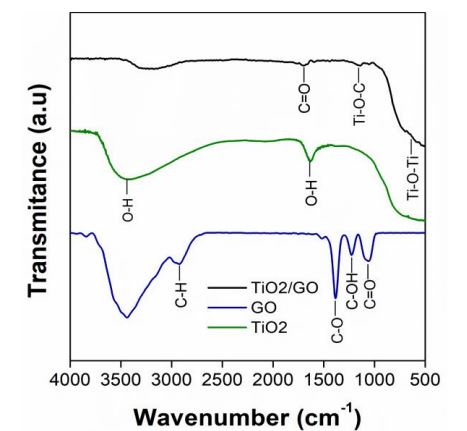
Figure 2 shows the FTIR spectra of TiO2, GO and the TiO2/GO composite obtained in this study. All the samples present a band between 3000 cm-1 and 3377 cm-1, which corresponds to the stretching vibration of the O-H bond. The spectrum of TiO2 shows a band around 1610 cm-1related to the bending vibration mode of the O-H bond, and characteristic bands of titanium dioxide are located at 400 cm-1 and 800 cm-1. The spectrum of GO shows several bands, but the one at 2939 cm-1 is due to C-H stretching vibration, which occurs during the synthesis process [
The thermal stability of the GO and TiO2/GO was examined by TGA. Figures 3a and 3b show the TGA and DTG results of each type of sample. The weight loss below 100 °C is due to the decomposition of interstitial water [
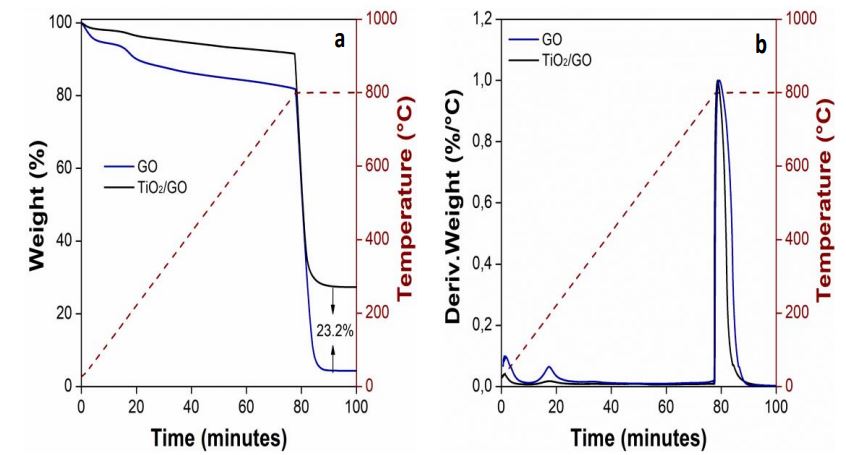
TGA and FTIR analyses confirmed the presence of oxygen-containing functional groups. These oxygen functionalities can be related to the oxidation of graphite by hydroxyl anions. The OH- anion attacks the sp. carbon atoms at the edge of graphite rods, causing the expansion of layers [
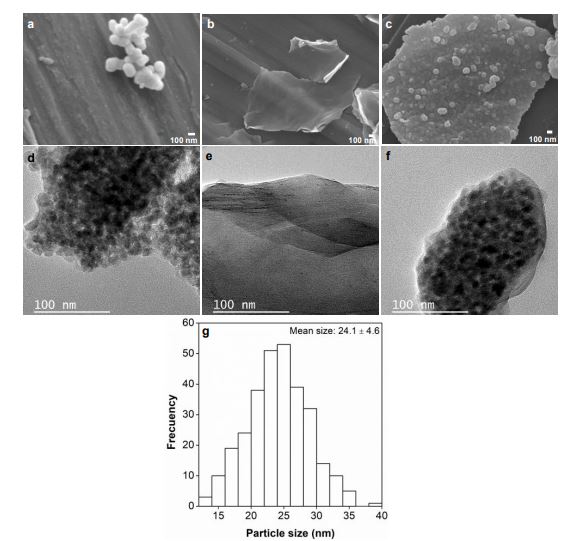
The morphology of the materials analyzed in this study was evaluated by SEM and TEM. Figures 4a, 4b, and 4c are SEM micrographs of TiO2, GO and the TiO2/GO composite, respectively. Figure 4a reveals that TiO2 nanoparticles have a spherical morphology. On the other hand, the micrograph of GO (Figure 4b) shows a thin graphene oxide layer; as a result, the background is visible. The micrograph of TiO2/GO (Figure 4c) presents TiO2 nanoparticles deposited on a GO layer. In addition, the TEM micrograph of GO (Figure 4f) confirms the exfoliation of graphite rods, which is a that involves the evolution of several gaseous species (SO2, O2, CO and CO2) between layers. These species come from the reduction of the SO4-2 anion, water oxidation and carbon corrosion [
The average diameter of the TiO2 particles in the SEM images was measured by ImageJ software using more than 250 nanoparticles. Figure 4g shows the mean particle size: 24.1 ± 4.6 nm.
Source: Own elaboration.
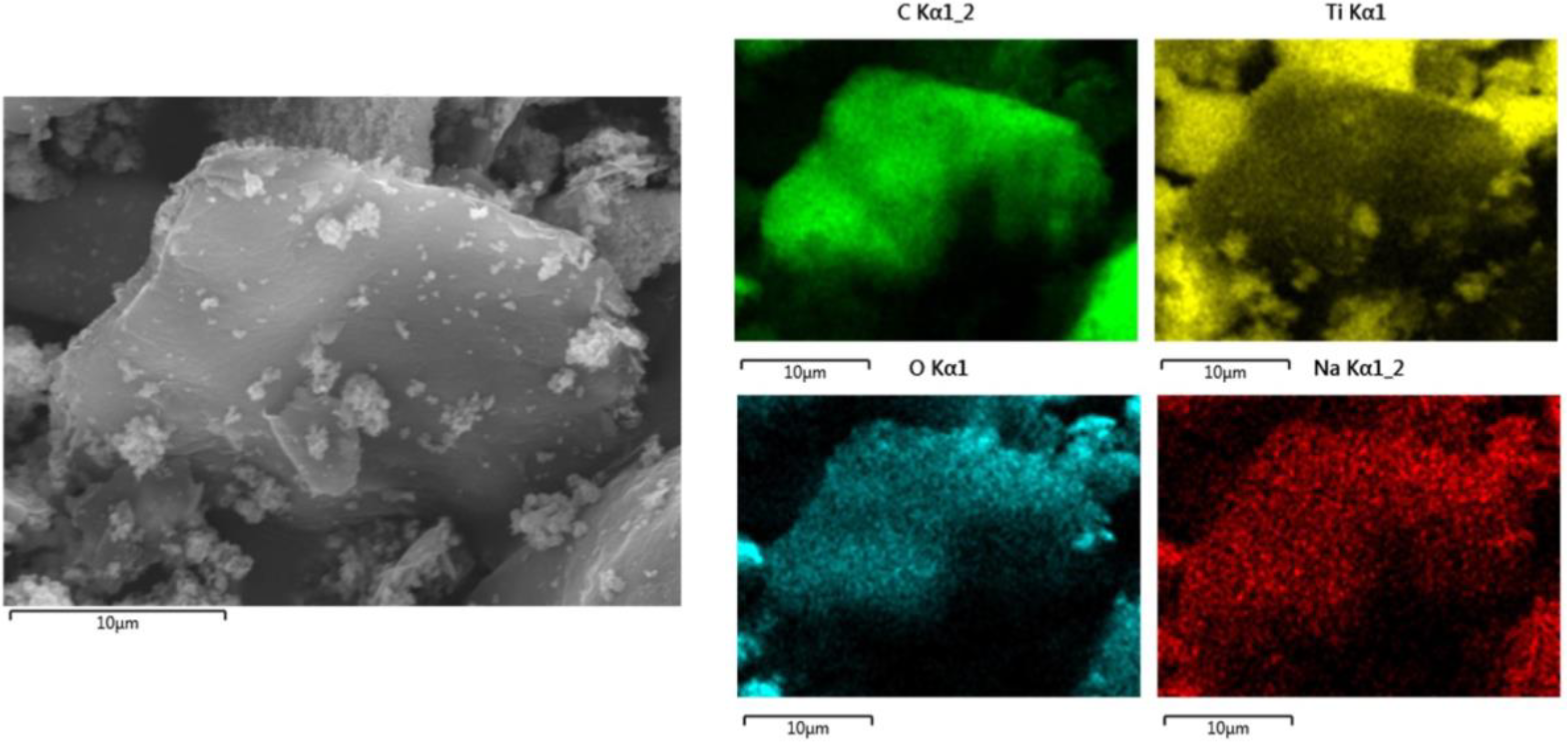
The chemical composition of the TiO2/GO composite was obtained by Energy Dispersive X-ray Spectroscopy (EDS). Figure 5 shows the EDS spectrum of the hybrid nanomaterial NM and Ti; O and C were found in the sample. Additionally, sodium (Na), which can also be observed in the spectrum, is related to remnants of electrolyte (Na2SO4) used during the synthesis of graphene oxide.
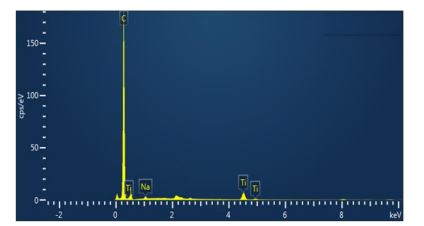
Figure 6 shows the element distribution map obtained from the TiO2/GO composite. The map identified the same elements found in the EDS spectrum (C, Ti, O and Na). In addition, the maps clearly show that the nanoparticles of titanium dioxide are located on the surface of the GO layers.
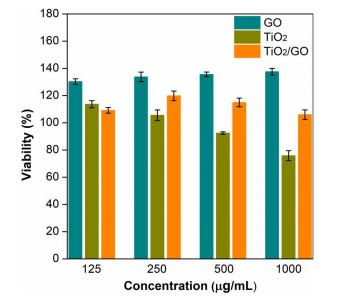
The antibacterial activity of the samples prepared here was determined by the macrodilution method. Figure 7 shows the bacterial viability of E. coli under different concentrations of GO, TiO2 and TiO2/GO. In the results, the graphene oxide sample presents an increase in the viability of E. Coli when the concentration of the treatment is higher.
Source: Own elaboration.
However, the TiO2 nanoparticles show an inverse trend, i.e., the viability of E. Coli decreases as the concentration of nanoparticles increases. At the lowest concentration (i.e., 125 µg/mL), the viability of the bacteria in the TiO2/GO hybrid material was 23 % and 17 % lower than in the GO and TiO2 samples, respectively. Nonetheless, at higher concentrations (250, 500 and 1000 µg / mL), the antibacterial activity increased in GO samples but decreased in samples containing TiO. nanoparticles. These results indicate that each treatment follows a different trend in terms of bactericidal activity as a function of concentration.
The antibacterial activity of graphene oxide (GO) reported in several papers is controversial. Nevertheless, such activity can be explained by three main mechanisms: membrane rupture, oxidative stress and isolation of bacteria envelope [
On the other hand, TiO2 nanoparticles affect biological systems since their photocatalytic activity generates potential reactive oxygen species (ROS) on their surfaces. ROS cause peroxidation of phospholipids in the cell membrane, inducing its breakdown [
Finally, the behavior of the composite material was similar to that reported by [
In addition, MIC50 is the minimum amount of drug/compound to inhibit 50 % of microorganism growth and was calculated. GO MIC50 could not be determined because bacterial viability does not decrease with increasing concentration. The MIC50 of TiO2/GO and TiO2 was 4575 µg/mL and 1728 µg/ mL, respectively, which confirms that TiO2 shows a higher bactericidal activity than GO and TiO2/GO.
4. CONCLUSIONS
This study assessed the effect of TiO2, GO, and TiO2/GO nanocompounds on bacterial activity against Escherichia coli. Using the hydrothermal method, TiO2 nanoparticles were obtained with an anatase phase and an average size of 24.1 ± 4.6 nm. Graphene oxide was obtained through electrochemical exfoliation, a simple and environmentally friendly method that induced a lower amount of oxygen-containing functional groups on the GO surface. A 23.2 % TiO2 content was evidenced in the developed hybrid nanomaterial. Furthermore, GO promoted bacterial growth due to its surface functional groups. TiO2 exhibited stronger bactericidal activity than the TiO2/GO compound. This behavior may be associated with the decreased contact between TiO2 and bacterial cells due to the blocking of active sites on the TiO2 surface by graphene sheets.
5. ACKNOWLEDGMENTS
The authors would like to thank the Polymers Laboratory and the Advanced Materials and Energy Research Group (MATyER) at the Instituto Tecnológico Metropolitano (Medellín, Colombia) for providing the chemical reagents and infrastructure used in this research.
CONFLICTS OF INTEREST
None of the authors have any conflict of interests regarding the publication of this work.
AUTHOR CONTRIBUTIONS
G. Durango-Giraldo, C. Zapata-Hernandez, C. Betancur-Henao. Contributed equally to this work. Roles: Investigation, Methodology, Validation, Writing – original draft.
J.F Santa-Marín and R. Buitrago-Sierra. Contributed equally to this work. Roles: Conceptualization, Formal analysis, Investigation, Methodology, Supervision, Validation, Writing – review & editing.
7. REFERENCES
- arrow_upward [1] H. A. Khan, A. Ahmad, and R. Mehboob, “Nosocomial infections and their control strategies,” Asian Pac. J. Trop. Biomed., vol. 5, no. 7, pp. 509–514, Jul. 2015. https://doi.org/10.1016/j.apjtb.2015.05.001
- arrow_upward [2] S. L. Percival, and D. W. Williams, “Escherichia coli,” in Microbiology of Waterborne Diseases, Elsevier, 2014, pp. 89–117. https://doi.org/10.1016/B978-0-12-415846-7.00006-8
- arrow_upward [3] E. P. Dellinger, “Prevention of Hospital-Acquired Infections,” Surg Infect (Larchmt)., vol. 17, no. 4, pp. 422–426, Jul. 2016. https://doi.org/10.1089/sur.2016.048
- arrow_upward [4] Z. Chao, W. Xinru, L. Aihui, C. Pan, H. Ding, and Y. Wei, “Reduced graphene oxide/titanium dioxide hybrid nanofiller-reinforced electrospun silk fibroin scaffolds for tissue engineering - ScienceDirect,” Mater. Lett., vol. 291, p. 129563, May. 2021. https://doi.org/10.1016/j.matlet.2021.129563
- arrow_upward [5] C. Ying-Na et al., “Synthesis of magnetic graphene oxide-TiO2 and their antibacterial properties under solar irradiation,” Appl. Surf. Sci., vol. 343, pp. 1–10, Jul. 2015. https://doi.org/10.1016/j.apsusc.2015.03.082
- arrow_upward [6] H. Mohammed et al., “Antimicrobial Mechanisms and Effectiveness of Graphene and Graphene-Functionalized Biomaterials. A Scope Review,” Front. Bioeng. Biotechnol., vol. 8, May. 2020. https://doi.org/10.3389/fbioe.2020.00465
- arrow_upward [7] H. M. Hegab, A. Elmekawy, L. Zou, D. Mulcahy, C. P. Saint, and M. Ginic-Markovic, “The controversial antibacterial activity of graphene-based materials,” Carbon N. Y., vol. 105, pp. 362–376, Aug. 2016. https://doi.org/10.1016/j.carbon.2016.04.046
- arrow_upward [8] J. Qiu, L. Liu, H. Zhu, and X. Liu, “Combination types between graphene oxide and substrate affect the antibacterial activity,” Bioactive Materials, vol. 3, no. 3, pp. 341–346, Sep. 2018. https://doi.org/10.1016/j.bioactmat.2018.05.001
- arrow_upward [9] A. A. Menazea, and M. K. Ahmed, “Synthesis and antibacterial activity of graphene oxide decorated by silver and copper oxide nanoparticles,” J. Mol. Struct., vol. 1218, p. 128536, Oct. 2020. https://doi.org/10.1016/j.molstruc.2020.128536
- arrow_upward [10] K. Zhu, H. Tian, X. Zheng, L. Wang, and X. Wang, “Triangular silver nanoparticles loaded on graphene oxide sheets as an antibacterial film,” Materials Letters, vol. 275, p. 128162, Sep. 2020. https://doi.org/10.1016/j.matlet.2020.128162
- arrow_upward [11] H. Feng, R. Cheng, X. Zhao, X. Duan, and J. Li, “A low-temperature method to produce highly reduced graphene oxide,” Nat. Commun., vol. 4, p.1539, Feb. 2013. https://doi.org/10.1038/ncomms2555
- arrow_upward [12] M. J. Fernández-Merino et al., “Vitamin C is an ideal substitute for hydrazine in the reduction of graphene oxide suspensions,” Journal of Physical Chemistry C., vol. 114, no. 14, pp. 6426–6432, Mar. 2010. https://doi.org/10.1021/jp100603h
- arrow_upward [13] C. Xu, X. Shi, A. Ji, L. Shi, C. Zhou, and Y. Cui, “Fabrication and characteristics of reduced graphene oxide produced with different green reductants,” PLoS One, vol. 10, no. 12, p. e0144842, Dec. 2015. https://doi.org/10.1371/journal.pone.0144842
- arrow_upward [14] V. Likodimos, “Photonic crystal-assisted visible light activated TiO2photocatalysis,” Appl. Catal. B: Environmental., vol. 230, pp. 269–303, Aug. 2018. https://doi.org/10.1016/j.apcatb.2018.02.039
- arrow_upward [15] C. Dette et al., “TiO2 Anatase with a Bandgap in the Visible Region,” Nano. Lett., vol. 14, no. 11, pp. 6533–6538, Sep. 2014. https://doi.org/10.1021/nl503131s
- arrow_upward [16] U. Diebold, “The surface science of titanium dioxide,” Surf. Sci. Rep., vol. 48, no. 5–8, pp. 53–229, Jan. 2003. https://doi.org/10.1016/S0167-5729(02)00100-0
- arrow_upward [17] A. J. Haider, Z. N. Jameel, and I. H. M. Al-Hussaini, “Review on: Titanium dioxide applications,” Energy Procedia, vol. 157, pp. 17–29, Jan. 2019. https://doi.org/10.1016/j.egypro.2018.11.159
- arrow_upward [18] T. Gakhar, and A. Hazra, “p-TiO2/GO heterojunction based VOC sensors: A new approach to amplify sensitivity in FET structure at optimized gate voltage,” Measurement, vol. 182, p. 109721, Sep. 2021. https://doi.org/10.1016/J.MEASUREMENT.2021.109721
- arrow_upward [19] Y. Sang et al., “Enhanced photocatalytic property of reduced graphene oxide/TiO2 nanobelt surface heterostructures constructed by an in situ photochemical reduction method,” Small, vol. 10, no. 18, pp. 3775–3782, Sep. 2014. https://doi.org/10.1002/SMLL.201303489
- arrow_upward [20] W. Fan, Q. Lai, Q. Zhang, and Y. Wang, “Nanocomposites of TiO2 and Reduced Graphene Oxide as Efficient Photocatalysts for Hydrogen Evolution,” Journal of Physical Chemistry C, vol. 115, no. 21, pp. 10694–10701, May. 2011. https://doi.org/10.1021/JP2008804
- arrow_upward [21] M. Karimi-Nazarabad, E. K. Goharshadi, and M. Aziznezhad, “Solar Mineralization of Hard-Degradable Amphetamine Using TiO2/RGO Nanocomposite,” ChemistrySelect, vol. 4, no. 48, pp. 14175–14183, Dec. 2019. https://doi.org/10.1002/SLCT.201903943
- arrow_upward [22] Y. Jia, C. Hu, P. Shi, Q. Xu, W. Zhu, and R. Liu, “Effects of cellulose nanofibrils/graphene oxide hybrid nanofiller in PVA nanocomposites,” Int. J. Biol. Macromol., vol. 161, pp. 223–230, Oct. 2020. https://doi.org/10.1016/J.IJBIOMAC.2020.06.013
- arrow_upward [23] I. Kartini, P. Meredith, J. C. D. Da Costa, and G. Q. Lu, “A novel route to the synthesis of mesoporous titania with full anatase nanocrystalline domains,” J. Solgel. Sci. Technol., vol. 31, no. 1-3, pp. 185–189, Aug. 2004. https://doi.org/10.1023/B:JSST.0000047984.60654.a1
- arrow_upward [24] G. Durango-Giraldo, A. Cardona, J. F. Zapata, J. F. Santa, and R. Buitrago-Sierra, “Titanium dioxide modified with silver by two methods for bactericidal applications,” Heliyon, vol. 5, no. 5, p. e01608, May. 2019. https://doi.org/10.1016/j.heliyon.2019.e01608
- arrow_upward [25] R. Boardman, and R. A. Smith, “Evaluating the efficacy of an essential oil extract of thyme (Thymus vulgaris) against methicillin-sensitive and methicillin-resistant strains of Staphylococci,” American Journal of Essential Oils and Natural Products, vol. 4, no. 2, pp. 17–22, Apr. 2016. https://www.essencejournal.com/pdf/2016/vol4issue2/PartA/4-2-3-902.pdf
- arrow_upward [26] C. Zapata-Hernandez, G. Durango-Giraldo, K. Cauca, and R. Buitrago-Sierra, “Influence of graphene oxide synthesis methods on the electrical conductivity of cotton/graphene oxide composites,” The Journal of The Textile Institute, vol. 113, no. 1, pp. 131-140, Dec. 2020. https://doi.org/10.1080/00405000.2020.1865507
- arrow_upward [27] X. Wei, G. Zhu, J. Fang, and J. Chen, “Synthesis, characterization, and photocatalysis of well-dispersible phase-pure anatase TiO2 nanoparticles,” International Journal of Photoenergy, vol. 2013, Apr. 2013. https://doi.org/10.1155/2013/726872
- arrow_upward [28] S. E. Bourdo et al., “Physicochemical characteristics of pristine and functionalized graphene,” Journal of Applied Toxicology, vol. 37, no. 11, pp. 1288–1296, Nov. 2017. https://doi.org/10.1002/jat.3493
- arrow_upward [29] Y. Z. N. Htwe, W. S. Chow, Y. Suda, A. A. Thant, and M. Mariatti, “Effect of electrolytes and sonication times on the formation of graphene using an electrochemical exfoliation process,” Applied Surface Science, vol. 469. pp. 951–961, Mar. 2019. https://doi.org/10.1016/j.apsusc.2018.11.029
- arrow_upward [30] K. K. De Silva, H. Hsin-Hui, S. Suzuki, and M. Yoshimura, “Ethanol-assisted restoration of graphitic structure with simultaneous thermal reduction of graphene oxide,” Jpn. J. Appl. Phys., vol. 57, no. 8S1, p. 08NB03, Jun. 2018. https://doi.org/10.7567/JJAP.57.08NB03
- arrow_upward [31] A. Ilnicka, M. Skorupska, P. Kamedulski, and J. P. Lukaszewicz, “Electro-exfoliation of graphite to graphene in an aqueous solution of inorganic salt and the stabilization of its sponge structure with poly(Furfuryl alcohol),” Nanomaterials, vol. 9, no. 7, p. 971, Jul. 2019. https://doi.org/10.3390/nano9070971
- arrow_upward [32] X. Mei, X. Meng, and F. Wu, “Hydrothermal method for the production of reduced graphene oxide,” Physica E: Low-Dimens Syst Nanostruct., vol. 68, pp. 81–86, Apr. 2015. https://doi.org/10.1016/j.physe.2014.12.011
- arrow_upward [33] R. Kumar et al., “Bulk synthesis of highly conducting graphene oxide with long range ordering,” RSC Adv., vol. 5, no. 45, pp. 35893–35898, Apr. 2015. https://doi.org/10.1039/c5ra01943e
- arrow_upward [34] K. Min-Sik, W. Jeong-Min, G. Dae-Myeong, J. R. Rani, and J. Jae-Hyung, “Effect of copper surface pre-treatment on the properties of CVD grown graphene,” AIP Adv., vol. 4, no. 12, Dec. 2014. https://doi.org/10.1063/1.4903369
- arrow_upward [35] J. R. Anasdass, P. Kannaiyan, R. Raghavachary, S. C. B. Gopinath, and Y. Chen, “Palladium nanoparticle-decorated reduced graphene oxide sheets synthesized using Ficus carica fruit extract: A catalyst for Suzuki cross-coupling reactions,” PLoS One, vol. 13, no. 2, p. e0193281, Feb. 2018. https://doi.org/10.1371/journal.pone.0193281
- arrow_upward [36] S. A. Khan et al., “Synthesis of TiO2/Graphene oxide nanocomposites for their enhanced photocatalytic activity against methylene blue dye and ciprofloxacin,” Compos. B. Eng., vol. 175, p. 107120, Oct. 2019. https://doi.org/10.1016/j.compositesb.2019.107120
- arrow_upward [37] T. Lling-Lling, O. Wee-Jun, C. Siang-Piao, and A. R. Mohamed, “Reduced graphene oxide-TiO2 nanocomposite as a promising visible-light-active photocatalyst for the conversion of carbon dioxide,” Nanoscale Res. Lett., vol. 8, no. 1, p. 465, Nov. 2013. https://doi.org/10.1186/1556-276X-8-465
- arrow_upward [38] K. K. Abbas, and A. M. H. A. Al-Ghaban, “Enhanced solar light photoreduction of innovative TiO2 nanospherical shell by reduced graphene oxide for removal silver ions from aqueous media,” Journal of Environmental Chemical Engineering, vol. 7, no. 3, p. 103168, Jun. 2019. https://doi.org/10.1016/j.jece.2019.103168
- arrow_upward [39] Y. Ren, L. Zhao, Y. Zou, L. Song, N. Dong, and J. Wang, “Effects of different TiO2 particle sizes on the microstructure and optical limiting properties of TiO2/reduced graphene oxide nanocomposites,” Nanomaterials, vol. 9, no. 5, p.730, May. 2019. https://doi.org/10.3390/nano9050730
- arrow_upward [40] M. P. Lavin-Lopez, A. Paton-Carrero, L. Sanchez-Silva, J. L. Valverde, and A. Romero, “Influence of the reduction strategy in the synthesis of reduced graphene oxide,” Advanced Powder Technology, vol. 28, no. 12, pp. 3195–3203, Dec. 2017. https://doi.org/10.1016/j.apt.2017.09.032
- arrow_upward [41] R. Larciprete, P. Lacovig, S. Gardonio, A. Baraldi, and S. Lizzit, “Atomic oxygen on graphite: Chemical characterization and thermal reduction,” Journal of Physical Chemistry C, vol. 116, no. 18, pp. 9900–9908, Apr. 2012. https://doi.org/10.1021/jp2098153
- arrow_upward [42] C. Botas et al., “Critical temperatures in the synthesis of graphene-like materials by thermal exfoliation-reduction of graphite oxide,” Carbon N. Y, vol. 52, p. 476-485, Feb. 2013. https://doi.org/10.1016/j.carbon.2012.09.059
- arrow_upward [43] G. Zhang, M. Wen, S. Wang, J. Chen, and J. Wang, “Insights into thermal reduction of the oxidized graphite from the electro-oxidation processing of nuclear graphite matrix,” RSC Adv., vol. 8, no. 1, pp. 567–579, Jan. 2018. https://doi.org/10.1039/c7ra11578d
- arrow_upward [44] V. Z. Baldissarelli, T. De Souza, L. Andrade, L. F. C. De Oliveira, H. J. José, and R. D. F. P. Muniz Moreira, “Preparation and photocatalytic activity of TiO2 -exfoliated graphite oxide composite using an ecofriendly graphite oxidation method,” Applied Surface Science, vol. 359, pp. 868–874, Dec. 2015. https://doi.org/10.1016/j.apsusc.2015.10.199
- arrow_upward [45] P. Yu, S. E. Lowe, G. P. Simon, and Y. L. Zhong, “Electrochemical exfoliation of graphite and production of functional graphene,” Curr. Opin. Colloid Interface Sci., vol. 20, no. 5–6, pp. 329–338, Oct-Dec. 2015. https://doi.org/10.1016/j.cocis.2015.10.007
- arrow_upward [46] L. Qiu et al., “Dispersing carbon nanotubes with graphene oxide in water and synergistic effects between graphene derivatives,” Chemistry - A European Journal, vol. 16, no. 35, pp. 10653–10658, Sep. 2010. https://doi.org/10.1002/chem.201001771
- arrow_upward [47] N. Keklikcioglu Cakmak, “The impact of surfactants on the stability and thermal conductivity of graphene oxide de-ionized water nanofluids,” J. Therm. Anal. Calorim., vol. 139, no. 3, pp. 1895–1902, Dec. 2020. https://doi.org/10.1007/s10973-019-09096-6
- arrow_upward [48] H. Lee, J. I. Choi, J. Park, S. S. Jang, and S. W. Lee, “Role of anions on electrochemical exfoliation of graphite into graphene in aqueous acids,” Carbon N. Y., vol. 167, pp. 816–825, Oct. 2020. https://doi.org/10.1016/j.carbon.2020.06.044
- arrow_upward [49] M. Yousefi et al., “Anti-bacterial activity of graphene oxide as a new weapon nanomaterial to combat multidrug-resistance bacteria,” Materials Science and Engineering C Mater. Biol. Appl., vol. 74. pp. 568–581, May. 2017. https://doi.org/10.1016/j.msec.2016.12.125
- arrow_upward [50] N. S. Ahmad, N. Abdullah, and F. M. Yasin, “Toxicity assessment of reduced graphene oxide and titanium dioxide nanomaterials on gram-positive and gram-negative bacteria under normal laboratory lighting condition,” Toxicology Reports, vol. 7, pp. 693–699, 2020. https://doi.org/10.1016/j.toxrep.2020.04.015
- arrow_upward [51] A. Khan, F. Ameen, F. Khan, A. Al-Arfaj, and B. Ahmed, “Fabrication and antibacterial activity of nanoenhanced conjugate of silver (I) oxide with graphene oxide,” Mater. Today Commun., vol. 25, p. 101667, Dec. 2020. https://doi.org/10.1016/j.mtcomm.2020.101667
- arrow_upward [52] H. Zheng et al., “Antibacterial applications of graphene oxides: structure-activity relationships, molecular initiating events and biosafety,” Science Bulletin, vol. 63, no. 2. pp. 133–142, Jan. 2018. https://doi.org/10.1016/j.scib.2017.12.012
- arrow_upward [53] C. Xie et al., “Elucidating the origin of the surface functionalization - dependent bacterial toxicity of graphene nanomaterials: Oxidative damage, physical disruption, and cell autolysis,” Science of the Total Environment, vol. 747, p. 141546, Dec. 2020. https://doi.org/10.1016/j.scitotenv.2020.141546
- arrow_upward [54] T. Zhang, and T. Pier-Luc, “Graphene: An Antibacterial Agent or a Promoter of Bacterial Proliferation?,” iScience, vol. 23, no. 12, p. 101787, Dec. 2020. https://doi.org/10.1016/j.isci.2020.101787
- arrow_upward [55] H. Luo, H. Ao, M. Peng, F. Yao, Z. Yang, and Y. Wan, “Effect of highly dispersed graphene and graphene oxide in 3D nanofibrous bacterial cellulose scaffold on cell responses: A comparative study,” Mater. Chem. Phys., vol. 235, p. 121774, Sep. 2019. https://doi.org/10.1016/j.matchemphys.2019.121774
- arrow_upward [56] A. Raja C. et al., “Decoration of 1-D nano bioactive glass on reduced graphene oxide sheets: Strategies and in vitro bioactivity studies,” Materials Science and Engineering C Mater. Biol. Appl., vol. 90, pp. 85–94, Sep. 2018. https://doi.org/10.1016/j.msec.2018.04.040
- arrow_upward [57] O. N. Ruiz et al., “Graphene oxide: a nonspecific enhancer of cellular growth,” ACS Nano, vol. 5, no. 10, pp. 8100–8107, Sep. 2011. https://doi.org/10.1021/nn202699t
- arrow_upward [58] V. Scuderi et al., “Photocatalytic and antibacterial properties of titanium dioxide flat film,” Mater. Sci. Semicond. Process., vol. 42, pp. 32–35, Feb. 2016. https://doi.org/10.1016/j.mssp.2015.09.005
- arrow_upward [59] Y. H. Leung et al., “Toxicity of ZnO and TiO2 to Escherichia coli cells.,” Sci. Rep., vol. 6, p. 35243, Oct. 2016. https://doi.org/10.1038/srep35243
- arrow_upward [60] A. Wanag et al., “Antibacterial properties of TiO2 modified with reduced graphene oxide,” Ecotoxicology and Environmental Safety, vol. 147, pp. 788–793, Jan. 2018. https://doi.org/10.1016/j.ecoenv.2017.09.039
 PDF
PDF

